FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors | Semantic Scholar
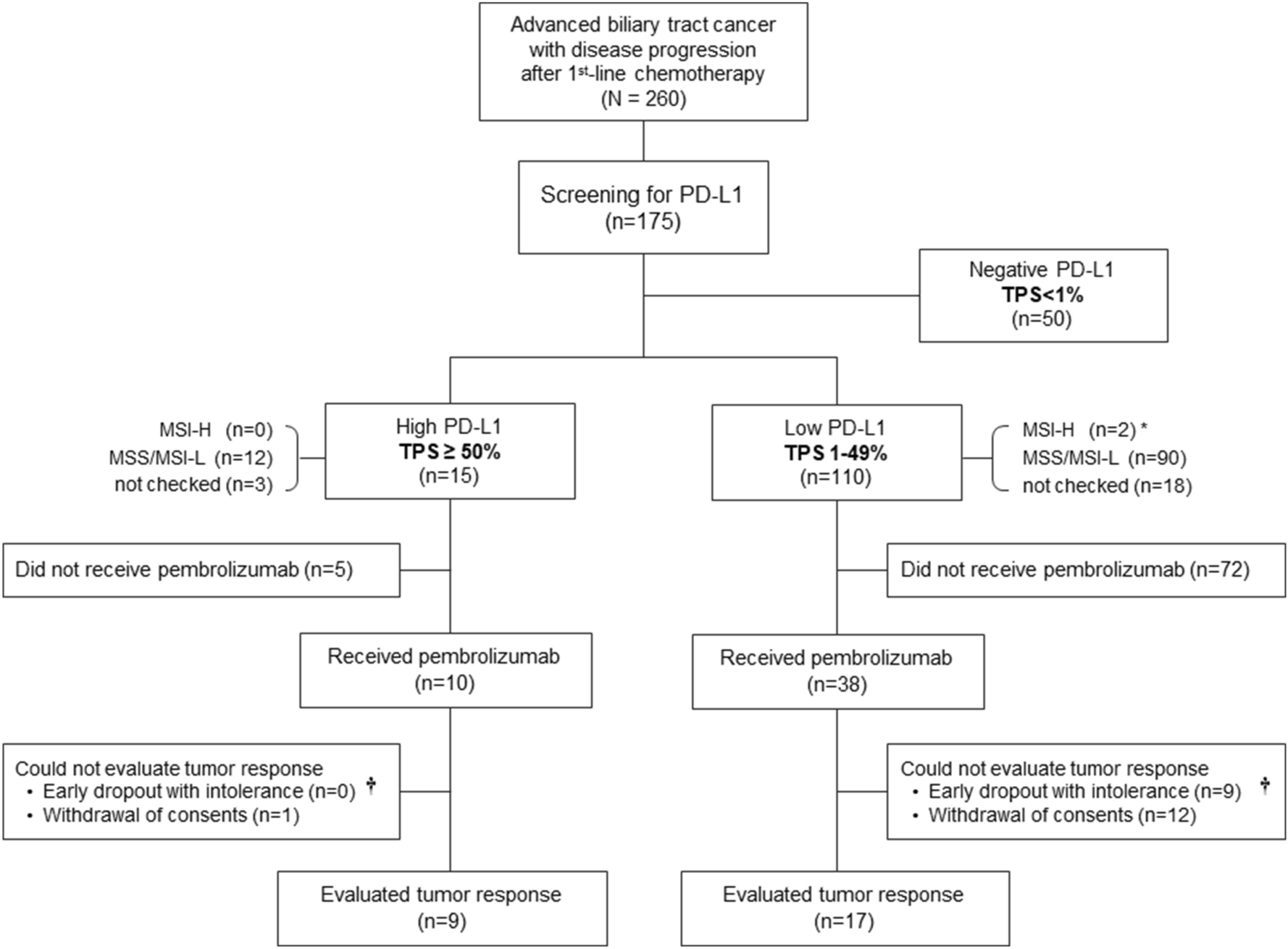
High PD-L1 expression is associated with therapeutic response to pembrolizumab in patients with advanced biliary tract cancer | Scientific Reports
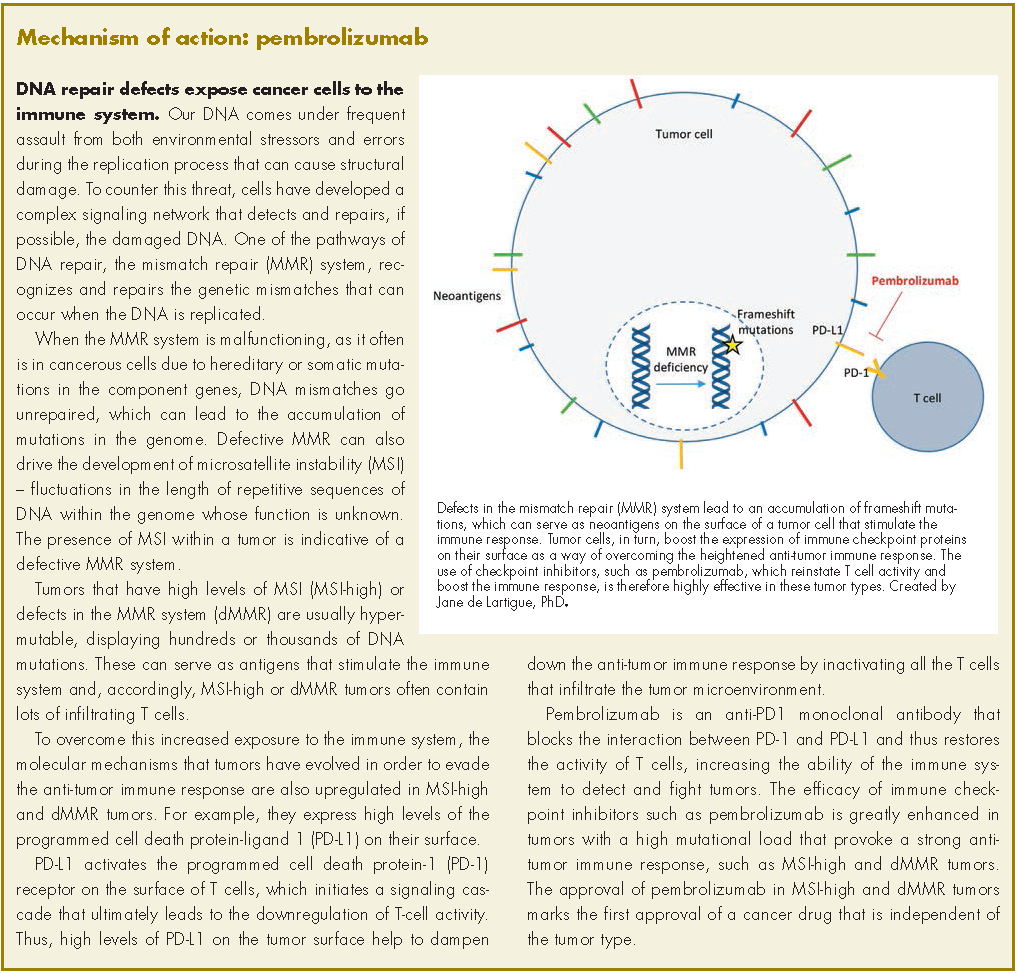
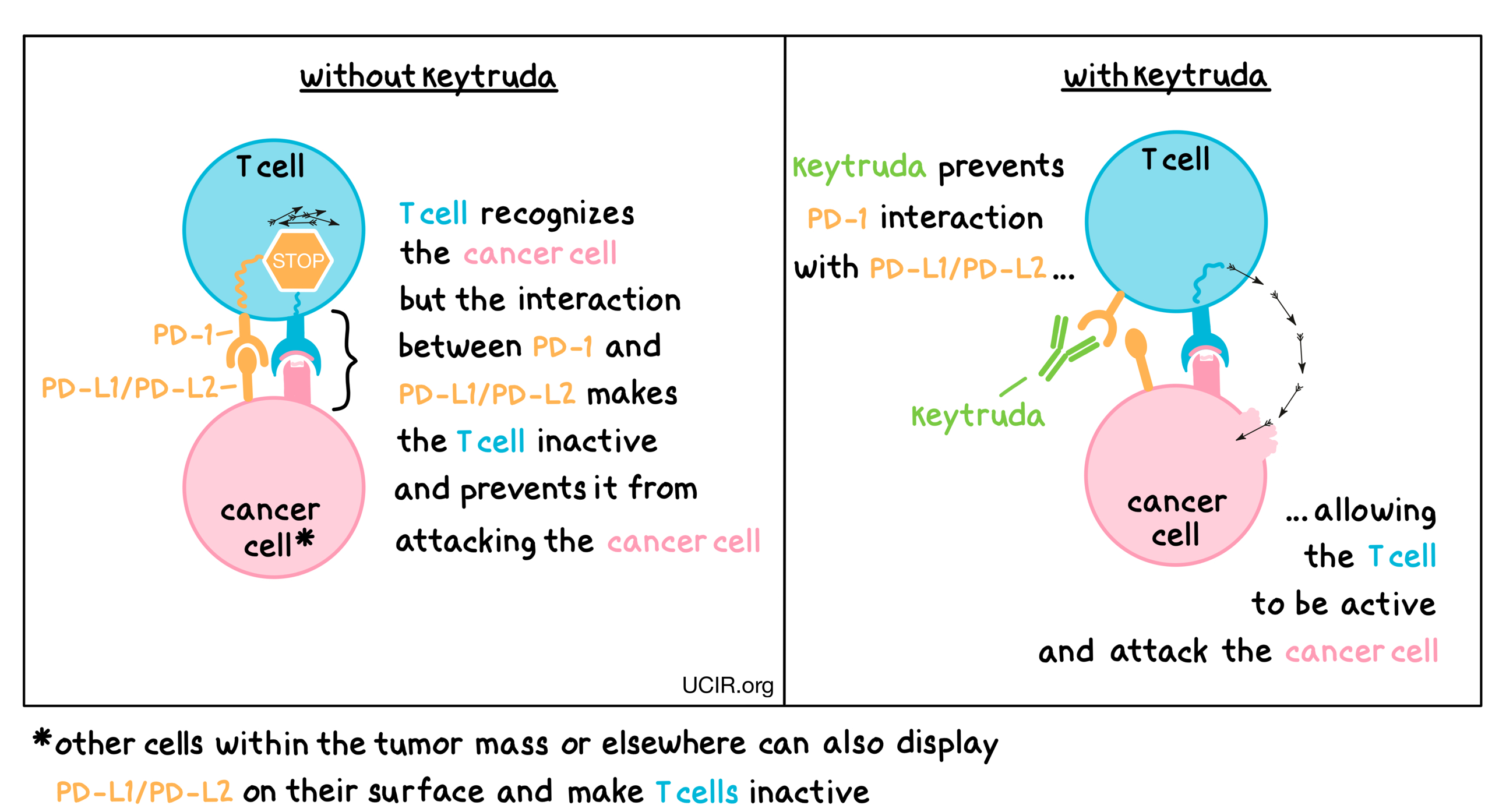
Pembrolizumab for dMMR/MSI-H tumors marks first tumor agnostic FDA approval | MDedge Hematology and Oncology
Approval of pembrolizumab (MSI-H/dMMR) and considerations for site-agnostic development of drugs in oncology

Pembrolizumab in microsatellite instability high or mismatch repair deficient cancers: updated analysis from the phase II KEYNOTE-158 study - Annals of Oncology

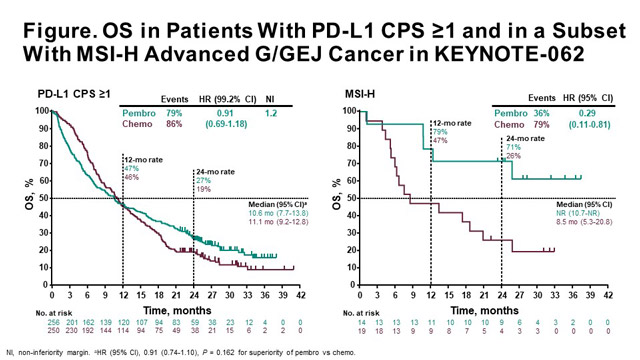
ESMO 2022: Pembrolizumab in microsatellite instability-high (MSI-H)/mismatch repair deficient (dMMR) advanced solid tumors: An update of the phase II KEYNOTE-158 trial

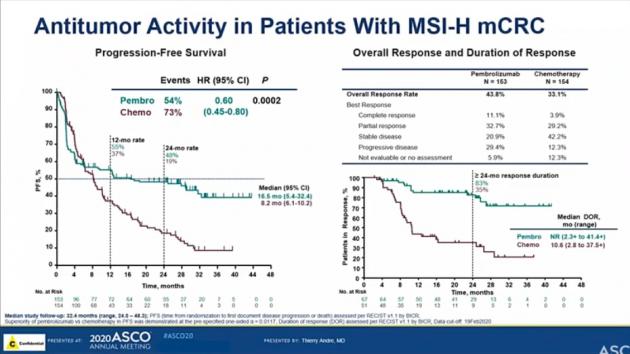
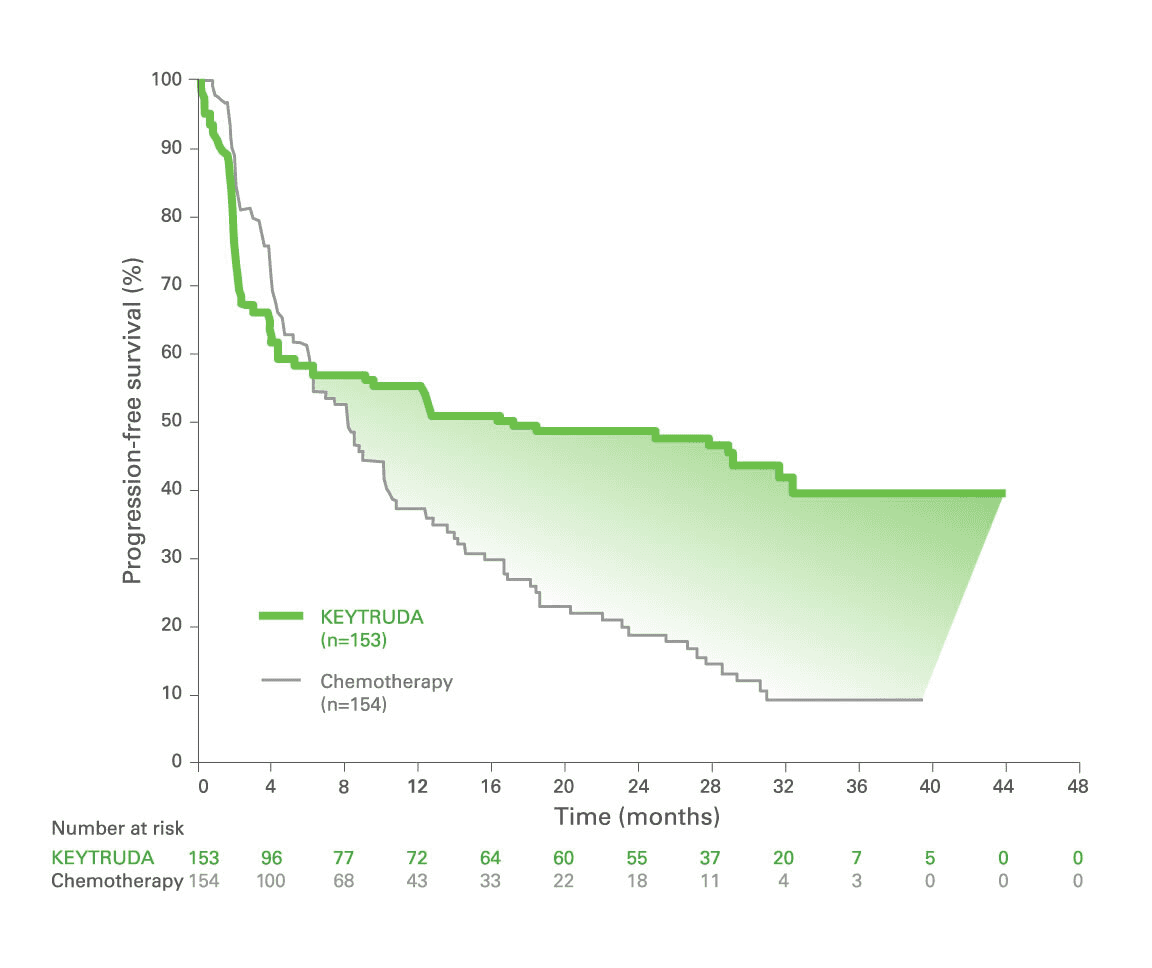
Study of Pembrolizumab (MK-3475) vs Standard Therapy in Participants With Microsatellite Instability-High (MSI-H)

Microsatellite instability and immune checkpoint inhibitors: toward precision medicine against gastrointestinal and hepatobiliary cancers | SpringerLink
Approval of pembrolizumab (MSI-H/dMMR) and considerations for site-agnostic development of drugs in oncology

FDA Converts to Full Approval Indication for KEYTRUDA® (pembrolizumab) for Certain Adult and Pediatric Patients With Advanced Microsatellite Instability-High (MSI-H) or Mismatch Repair Deficient (dMMR) Solid Tumors | Business Wire